Top Validation Frameworks for Healthcare AI Systems
Post Summary
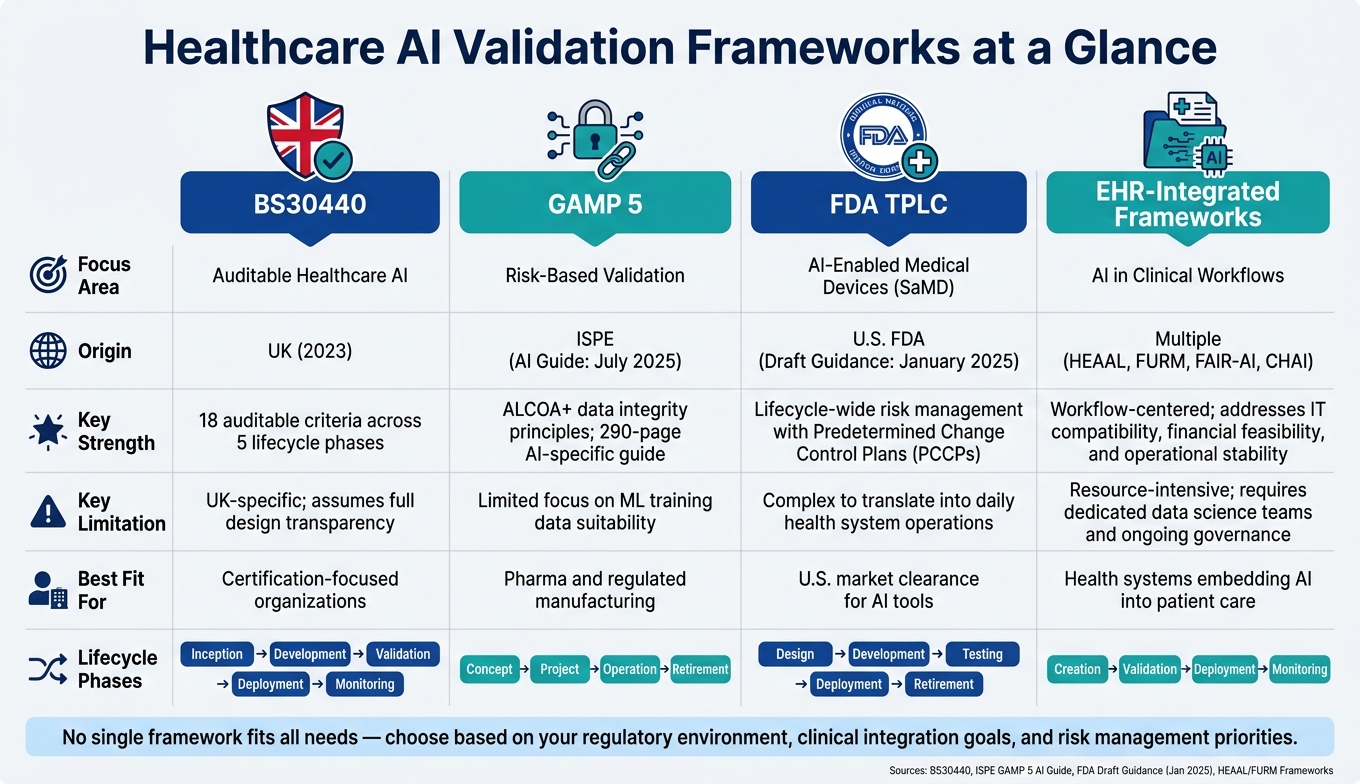
AI tools in healthcare offer immense potential, but without proper validation, they can lead to serious risks like misdiagnosis or unequal care. These failures often stem from disruptions to clinical applications and medical devices. Validation frameworks ensure these tools are safe, reliable, and effective. Here are four key frameworks for healthcare AI:
- BS30440: A UK-origin standard focusing on auditable criteria across five lifecycle phases. It emphasizes safety, clinical risk management, and ongoing monitoring.
- GAMP 5: A risk-based approach with strong data integrity measures and lifecycle phases tailored for AI systems, including dynamic model management.
- FDA's TPLC: A U.S.-specific framework ensuring continuous oversight of AI-enabled medical devices, from design to retirement, with a focus on risk management and real-world performance.
- EHR-Integrated Frameworks: Tailored for AI tools embedded in Electronic Health Records, addressing clinical workflow integration, safety, and data quality.
Quick Comparison:
| Framework | Focus Area | Strengths | Limitations | Best Fit For |
|---|---|---|---|---|
| BS30440 | Auditable healthcare AI | Structured phases, clear safety standards | UK-specific, assumes full transparency | Certification-focused organizations |
| GAMP 5 | Risk-based validation | Strong data integrity, lifecycle phases | Limited focus on training data quality | Pharma and regulated manufacturing |
| FDA's TPLC | AI-enabled medical devices | Lifecycle-wide risk management | Complex for daily operations | U.S. market clearance for AI tools |
| EHR-Integrated | AI in clinical workflows | Workflow-centered, safety-focused | Resource-intensive, governance-heavy | AI embedded in patient care workflows |
Each framework serves different needs, so choose based on your organization's goals - whether it's regulatory compliance, clinical integration, or long-term risk management.
Healthcare AI Validation Frameworks Compared: BS30440 vs GAMP 5 vs FDA TPLC vs EHR-Integrated
Evaluating AI Scribes: Frameworks for Safe and Reliable Summarization
sbb-itb-535baee
1. British Standard BS30440
Introduced in 2023, BS30440 stands as the first British Standard framework dedicated to auditable healthcare AI. Though developed in the UK, its structured approach to AI validation is gaining traction among U.S. healthcare organizations seeking a rigorous, globally-informed standard. This aligns with the projection that the global healthcare AI market will surpass $187.95 billion by 2030 [5].
Lifecycle Coverage
BS30440 organizes validation into a flexible five-phase lifecycle: inception, development, validation, deployment, and monitoring. This setup allows teams to revisit earlier phases when new risks or evidence arise. The framework incorporates 18 auditable assessment criteria across all phases, offering developers and procurement teams a clear checklist instead of vague recommendations. Engaging internal quality assurance managers early - during the inception phase - can help gather evidence upfront and minimize rework during audits [4]. This structured approach provides a solid foundation for managing risks and ensuring safety.
Clinical Risk and Safety Management
The framework takes clinical risk seriously by aligning with established safety standards like DCB 0129 and DCB 0160, which govern clinical risk management for medical devices and health IT systems [4][6]. It emphasizes more than just compliance; it requires active involvement from patients, users, and stakeholders throughout the lifecycle to reduce the likelihood of failures when transitioning from testing to real-world clinical use. Healthcare providers can even set BS30440 certification as a procurement requirement, ensuring a consistent safety benchmark across their vendors [4]. This process is a core component of effective third-party risk management in healthcare.
For instance, Lewisham and Greenwich NHS Trust applied BS30440 during a 2025 evaluation of an AI-powered fracture detection tool for emergency radiology. Led by Dr. Sarojini David, the team conducted shadow mode testing and ethical impact assessments. The selected tool demonstrated 97% accuracy, 93% sensitivity, and 98.8% specificity [6].
"Applying the BS 30440 framework in our comparative AI study has been transformative: not only in selecting the most clinically viable fracture detection tool for our service, but also in aligning our adoption process with safety, ethical, and regulatory best practices." - Dr. Sarojini David, Clinical Director of Radiology, AI Lead, Lewisham and Greenwich NHS Trust [6]
Continuous Monitoring and Adaptability
The monitoring phase is a critical component of BS30440. It requires ongoing performance tracking after deployment to identify issues like model drift, safety concerns, or changes in the patient population the AI was initially trained on [4]. For tools built on generic large language models (LLMs), the standard insists on an "assurance wrapper" around the underlying model to ensure compliance, even if the core algorithm wasn't developed by the supplier [4]. This requirement is particularly relevant today, as many healthcare AI tools rely on third-party foundation models.
2. GAMP 5 (Good Automated Manufacturing Practice)
GAMP 5's approach to risk-based validation fits seamlessly into the needs of healthcare AI. It relies on two primary documents: the GAMP 5 Second Edition, which outlines the core risk-based philosophy, and the ISPE GAMP Guide: Artificial Intelligence (released in July 2025). This guide, a comprehensive 290-page document created by over 20 industry experts, focuses specifically on AI systems [7].
Lifecycle Coverage
GAMP 5 reimagines the traditional V-model into four distinct phases: Concept, Project, Operation, and Retirement [7][9]. Here's what each phase entails:
- Concept Phase: Establishes the system's intended use and classifies it as either static or dynamic.
- Project Phase: Prioritizes data governance and evaluates potential biases.
- Operation Phase: Focuses on continuous monitoring to ensure ongoing system reliability.
- Retirement Phase: Requires archiving all relevant artifacts for the entire regulatory retention period.
This phased structure ensures that data controls remain stringent throughout the system's lifecycle.
Data Integrity and Security
To maintain high standards, GAMP 5 enforces compliance with ALCOA+ principles. These principles ensure that data remains Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, and Available [10][11]. Training, validation, and test datasets are tightly controlled with versioning to guarantee reproducibility and avoid contamination.
The 2025 AI Guide also addresses AI-specific risks, like data poisoning (where training data is tampered with), prompt injection (targeting large language models), and model exfiltration. As one expert explained:
"Annex 11 and Part 11 still apply, but now we must extend their controls into model training pipelines, cloud platforms, and retraining events." - Korrapati [9]
For compliance with 21 CFR Part 11, AI outputs cannot be independently signed by the model. Instead, a human reviewer must verify the output and apply their unique electronic signature [8].
Continuous Monitoring and Adaptability
Ongoing monitoring plays a critical role in GAMP 5's framework. During the Operation phase, real-time dashboards and automated drift detection systems are essential. These tools help identify performance drops and trigger retraining when necessary [10][9]. For dynamic models, it's vital to define adaptation boundaries early on. These boundaries specify the conditions under which a model can retrain without needing a complete re-qualification [7].
As ClinStacks emphasized:
"You cannot validate an AI system in February and assume it is still validated in November without ongoing performance and drift monitoring evidence." - ClinStacks [7]
3. FDA's Total Product Lifecycle (TPLC) Approach for SaMD

The FDA's Total Product Lifecycle (TPLC) framework is designed to address the unique challenges of AI-enabled medical devices, setting them apart from traditional software. Instead of relying on a single validation step, this approach requires continuous oversight through collaborative risk management, starting from the initial design phase and extending all the way to the device's retirement. In January 2025, the FDA published draft guidance specifically tailored for AI-enabled device software functions, formalizing this lifecycle-wide approach to risk management [14].
This framework stands out when compared to BS30440 and GAMP 5. While those standards focus on process-specific methodologies, TPLC offers a broader, ongoing model for overseeing AI products throughout their entire lifecycle.
"The complex and dynamic processes involved in the development, deployment, use, and maintenance of AI technologies benefit from careful management throughout the medical product life cycle." - FDA [14]
Lifecycle Coverage
The FDA's approach aligns the Software Development Lifecycle (SDLC) with an AI Lifecycle (AILC), covering everything from planning to retirement. These concepts are further refined by the Digital Health Center of Excellence (DHCoE) based on feedback from the community [12].
"Modern Software Development Lifecycles (SDLCs) embody LCM principles, offering a structured framework for planning, designing, implementing, testing, integrating, deploying, maintaining, and eventually retiring software." [12]
This comprehensive lifecycle coverage ensures rigorous evaluation of risks and performance at every stage.
Clinical Risk and Safety Management
To address clinical risks, the FDA employs the IMDRF risk categorization framework, which classifies SaMD into four tiers based on the severity of the healthcare situation and the role of the AI's output (Treat/Diagnose, Drive Clinical Management, or Inform Clinical Management) [13].
| State of Healthcare Situation | Treat or Diagnose | Drive Clinical Management | Inform Clinical Management |
|---|---|---|---|
| Critical | Category IV | Category III | Category II |
| Serious | Category III | Category II | Category I |
| Non-serious | Category II | Category I | Category I |
| Source: IMDRF/FDA Risk Categorization Framework [13] |
Category IV presents the highest patient safety impact, while Category I has the lowest [13]. This tiered system directly influences the level of scrutiny required. For example, an AI tool diagnosing a critical condition undergoes far more rigorous validation than one offering recommendations for non-serious clinical decisions.
Data Integrity and Security
The TPLC framework places a strong emphasis on "Data Suitability," which involves early assessments of data quality, coverage, and provenance to identify potential biases [12]. This step is critical in ensuring that any issues are addressed before they can impact patient safety. As highlighted by the DHCoE:
"While this adaptability can enhance performance, it also poses significant risks, such as exacerbating biases in data or algorithms, potentially harming patients and further disadvantaging underrepresented populations." - Troy Tazbaz, Director, and John Nicol, PhD, Digital Health Center of Excellence (DHCoE) [12]
Manufacturers must use specialized tools to detect and mitigate bias during data preprocessing, ensuring that the AI model performs reliably across all patient groups, not just those well-represented in the training data [12].
Continuous Monitoring and Adaptability
Recognizing the adaptive nature of AI, the TPLC framework incorporates Predetermined Change Control Plans (PCCPs). These plans allow manufacturers to pre-authorize future updates to their AI models without requiring a new regulatory submission each time [14]. This marks a shift from static approvals to a more dynamic regulatory model suited for evolving AI technologies. Additionally, the framework mandates Operation & Monitoring and Real-World Performance Evaluation phases post-deployment. These phases are critical for identifying performance issues or emerging biases before they can negatively affect patient care [12].
4. EHR-Integrated AI Validation Frameworks
The FDA's Total Product Lifecycle (TPLC) framework sets the standard for regulated medical devices, but when it comes to AI tools embedded in Electronic Health Record (EHR) systems, a different approach is needed. These tools often operate outside traditional medical device regulations, so specialized frameworks have been created to address challenges like data quality, patient safety, and operational stability. Unlike broader frameworks like TPLC and BS30440, EHR-integrated models are designed to meet the practical demands of clinical workflows.
Lifecycle Coverage
EHR-integrated frameworks outline the entire lifecycle of an AI tool - from its creation and development to validation, deployment, and ongoing monitoring. Instead of duplicating the structure of BS30440, these frameworks adapt it specifically for EHR settings. Take the HEAAL (Health Equity Across the AI Lifecycle) framework, for example. Developed with input from 77 healthcare practitioners, it incorporates eight critical decision points to ensure fairness and reliability throughout the process [16].
Before any AI tool is embedded into a clinical workflow, local validation is required to confirm it functions effectively in the provider's specific environment - not just in controlled lab conditions [1][15].
Clinical Risk and Safety Management
While traditional frameworks emphasize safety in controlled settings, EHR-integrated frameworks address the unpredictable nature of real-world clinical environments. The WHO framework, for instance, mandates clear performance goals and a forward-looking analysis plan to assess the AI's impact on clinical pathways before deployment [15].
"Transparently communicating clinical evaluation results is vital for safe and effective use of AI health technologies involving datasets, model descriptions, clinical studies, and post-deployment audits." - WHO Framework Mapping [15]
Human factors also play a crucial role. Frameworks like BS30440 emphasize the importance of ergonomics and clinician usability throughout the AI tool's lifecycle. Ensuring that healthcare providers can effectively interact with these systems is key to maintaining patient safety [4].
Data Integrity and Security
Building on safety concerns, these frameworks place a strong emphasis on maintaining data integrity. Protecting patient data and Personal Health Information (PHI) is a priority. EHR-integrated AI frameworks align with established safety standards, such as the UK's NHS Digital DCB 0129 and DCB 0160, to ensure data quality and security [4]. Additionally, the Coalition for Health AI (CHAI) is working on Testing and Evaluation frameworks for high-stakes EHR applications, like sepsis risk prediction and summarizing patient discharge notes [17].
Legal and regulatory risk assessments are also part of the process, ensuring compliance with privacy laws before integration [1][15]. For organizations leveraging foundation models like large language models (LLMs), frameworks recommend creating "assurance wrappers." These compliance layers uphold safety standards even when the model's internal workings are not fully transparent [4]. In EHR settings, where clinical stakes are high, this is particularly important. Tools like Censinet RiskOps™ provide structured workflows to help manage these AI-related risks effectively.
Continuous Monitoring and Adaptability
After deployment, continuous monitoring is critical. Real-time drift detection helps identify when models need updates or should be decommissioned, preventing disruptions caused by underperforming systems [1][15]. The FURM (Fair, Useful, and Reliable AI Models) framework takes this further by focusing on the real-time interaction between AI outputs and clinical decision-making [18][19].
"Estimating the effects of this interplay before deployment and studying it in real time after deployment are essential for bridging the chasm between AI model development and achievable benefits." - Stanford Law School [18]
Pros and Cons of Each Framework
Each framework comes with its own set of strengths and challenges, shaping how effectively it supports audit processes and ensures patient safety. Let’s break down the key points for each.
BS30440 consolidates scattered guidance into 18 auditable criteria, covering both clinical and non-clinical AI. It extends its scope to areas like logistics and resource planning and even includes considerations like carbon impact. However, it assumes that suppliers fully understand how their models were developed, which can be a major hurdle for opaque systems. Additionally, it is currently a UK-specific standard, which limits its applicability for international use, including U.S. regulatory contexts [4].
GAMP 5 focuses on data integrity through its ALCOA+ principles, ensuring consistency throughout the lifecycle. While this is a strong point, it falls short in evaluating the suitability of data used for training machine learning models. As noted in npj Digital Medicine:
"The quality of its training data... has fundamental impact on the resulting system. If the data used for training a model is bad, the resulting AI will be bad as well ('garbage in, garbage out')." - npj Digital Medicine [20]
This gap becomes especially critical for organizations deploying adaptive AI systems in regulated environments.
FDA's Total Product Lifecycle (TPLC) Approach aligns with rigorous transparency requirements for SaMD in the U.S. However, health systems often find it challenging to translate these guidelines into daily operations due to the context-specific nature of clinical risks. This disconnect can slow down the safe adoption of AI and increase compliance and vendor risk challenges.
EHR-Integrated Frameworks, like FAIR-AI and FURM, embed AI within clinical workflows, addressing practical concerns like IT compatibility, financial feasibility, and operational stability. However, they demand significant resources, including dedicated data science teams and active governance structures. These frameworks also require ongoing updates to keep pace with technological advancements. For instance, a study at Stanford Health Care found that only 2 out of 6 AI solutions assessed under the FURM framework moved forward to implementation [19]. Despite these challenges, these frameworks remain a go-to choice for health systems aiming to integrate AI directly into patient care.
Here’s a summary of the trade-offs:
| Framework | Primary Strength | Key Limitation | Best Use Case |
|---|---|---|---|
| BS30440 | Comprehensive and auditable; covers clinical and non-clinical AI [4] | Assumes full design transparency; UK-specific [4] | Organizations seeking formal certification |
| GAMP 5 | Strong focus on data integrity through ALCOA+ principles [20] | Lacks focus on data suitability for ML training [20] | Regulated manufacturing and pharma environments |
| FDA TPLC | Satisfies strict U.S. transparency needs for SaMD [2] | Limited practical guidance for health systems [2] | Developers aiming for U.S. market clearance |
| EHR-Integrated | Workflow-centered, scalable, and considers financial metrics [2][19] | Resource-intensive; requires ongoing governance [2] | Health systems embedding AI into clinical care |
Conclusion
The analysis highlights that each framework comes with its own strengths and limitations. No single option can address every need. Selecting the right framework depends on your organization’s focus - whether that’s regulatory compliance, seamless integration into clinical workflows, or managing long-term risks. This variety in validation approaches calls for a customized compliance strategy tailored to U.S. health systems.
For U.S. healthcare organizations, aligning with baseline regulatory standards is the first step. The HTI-1 transparency requirements are particularly important in this context. With ONC-certified health IT already supporting over 96% of hospitals and 78% of office-based physicians, most organizations are already operating under these guidelines [3]. Adding the NIST AI Risk Management Framework to this foundation provides a structured approach to managing broader risks. Notably, its April 2026 update will include a profile specifically designed for trustworthy AI in critical sectors like healthcare [21]. For organizations embedding AI into EHR workflows, compliance with USCDI v3 - set to become the baseline for ONC certification on January 1, 2026 - ensures interoperability and helps reduce data quality issues that could lead to bias [3].
In light of the framework comparisons, adhering to established standards emerges as essential for managing risks over the long term. For those needing to showcase accountability to external stakeholders, URAC Health Care AI Accreditation is gaining traction as a reliable indicator of governance maturity. As URAC explains:
"URAC Accreditation affirms conformance with URAC process and governance standards." [22]
FAQs
How do I choose the right validation framework for our AI tool?
To select the best validation framework, focus on clinical performance, safety, and adherence to healthcare standards. Consider frameworks like NIST Trustworthy AI, which emphasizes safety and privacy, BS30440, which addresses fairness and effectiveness, and FAIR-AI, designed for responsible implementation. Make sure the framework you choose aligns with your specific use case, regulatory requirements, and operational goals. Taking a well-rounded approach helps ensure AI is deployed safely, effectively, and in compliance with healthcare guidelines.
What should we monitor after an AI model goes live in the clinic?
After deployment, keep a close eye on the AI model's performance and accuracy by evaluating its outputs against clinical outcomes or established benchmarks. Be mindful of model drift, which can occur when data patterns or environments change over time. It's crucial to ensure the model's decisions stay safe, reliable, and in line with both ethical considerations and regulatory guidelines. Collecting feedback from clinicians and patients plays a key role in spotting usability issues and fostering trust. Make it a priority to regularly review how the model affects patient safety and the overall quality of care.
Do EHR-embedded AI tools require different validation than SaMD?
EHR-integrated AI tools share many validation principles with Software as a Medical Device (SaMD), but there are some key distinctions. Both need to prove clinical validity, safety, and effectiveness. However, tools embedded within electronic health record systems also have to tackle challenges like interoperability and ensuring smooth data flow within these systems. While the overall validation framework is similar, EHR tools require extra attention to these integration-specific aspects.


