Clinical Decision Support Systems (CDSS) are transforming healthcare by aiding physicians in making timely and informed decisions. However, these systems come with risks that can impact patient safety, including:
To mitigate these risks, healthcare organizations must evaluate vendors thoroughly, validate system performance across diverse populations, and implement continuous monitoring. Tools like Censinet RiskOps™ streamline risk assessments, combining automation with human oversight to ensure safety and reliability in CDSS deployment.
Key Takeaway: Without proper safeguards, CDSS risks - bias, inaccuracies, and cybersecurity vulnerabilities - can jeopardize patient care. Rigorous vendor evaluation and ongoing oversight are essential to minimize these risks and enhance trust in healthcare technology.
CDSS Vendor Risk Statistics: Medication Errors and Healthcare Costs
Main Vendor Risks in Clinical Decision Support Systems
When healthcare organizations rely on vendor-developed Clinical Decision Support Systems (CDSS), they expose themselves to challenges that can directly influence patient care and the overall quality of healthcare services.
Bias and Algorithm Fairness
One of the biggest issues with CDSS is algorithmic bias, which can undermine the system's reliability. When the training data reflects societal biases or lacks diversity, the system may unintentionally reinforce disparities. This can result in varying performance across demographic groups, potentially leading to incorrect or unsafe medical guidance for certain populations [2].
For instance, biased algorithms may provide misleading advice, suggest inappropriate treatments, or limit access to reliable information - particularly for minority groups or individuals with limited health literacy. Professor Anjana Susarla from Michigan State University highlights this concern:
The same algorithm will steer a less literate user toward fake cures or misleading medical advice. This could be especially harmful for minority groups
.
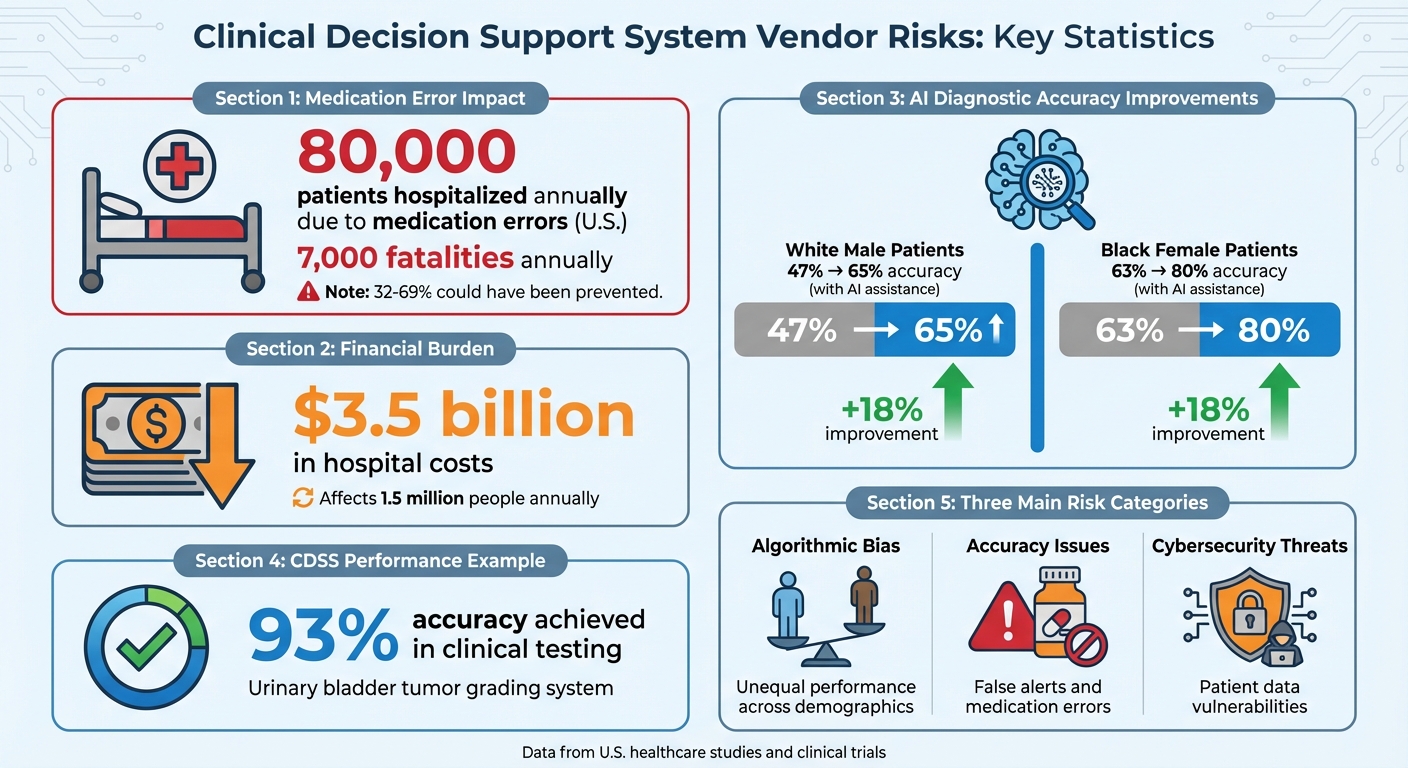
However, there are promising developments. A 2025 study published in Communications Medicine examined how AI assistance, specifically GPT-4, impacted physicians' diagnostic accuracy. In this study, 50 U.S.-licensed physicians reviewed chest pain scenarios involving a white male patient and a Black female patient. With AI support, diagnostic accuracy improved from 47% to 65% for white male patients and from 63% to 80% for Black female patients - an 18% improvement for both groups [2]. The study concluded:
Physician clinical decision making can be augmented by AI assistance while maintaining equitable care across patient demographics
.
While advances like this are encouraging, the risk of inaccuracies in CDSS remains a critical concern.
Accuracy and Clinical Safety
Even well-designed CDSS can generate false alerts or inconsistencies when integrated with electronic patient medical record (ePMR) systems, which increases the likelihood of medication errors [3]. In the U.S. alone, approximately 80,000 patients are hospitalized each year due to medication errors, with around 7,000 fatalities - 32% to 69% of which could have been prevented [3].
These errors not only jeopardize patient safety but also create significant financial strain. Medication-related mistakes affect an estimated 1.5 million people annually, adding $3.5 billion in hospital costs [3]. These figures underscore the need for stringent accuracy standards in CDSS to ensure both safety and cost-efficiency in healthcare.
How to Evaluate Vendor CDSS Risks
Healthcare organizations must adopt a thorough and structured approach to evaluate Clinical Decision Support System (CDSS) vendors. This process is crucial not only for safeguarding patients but also for reducing potential legal and operational risks. Below are key areas to focus on, from the initial stages of vendor selection to ongoing oversight.
Pre-Implementation Validation
Begin by requesting comprehensive validation reports from vendors. These reports should demonstrate the system's performance across a variety of patient populations. For instance, one CDSS designed for grading urinary bladder tumors and predicting recurrence achieved an impressive 93% accuracy during clinical testing [4]. Vendors should provide this level of detail as standard practice.
For AI-based CDSS, demand transparency regarding the algorithm's logic and the quality of training data. Diverse and high-quality datasets are essential to avoid the pitfalls of opaque, "black box" systems.
Also, evaluate how effectively the system prioritizes critical alerts while minimizing unnecessary notifications. Using high-priority drug–drug interaction lists as a benchmark can help ensure the system focuses on the most relevant risks.
Finally, consider how clinicians will integrate the system into their daily workflows. Research shows:
errors can still occur when providers bypass or misuse the technology
.
Your validation process should account for these real-world usage patterns to ensure the system performs reliably in practice.
Lifecycle Risk Management
Risk evaluation shouldn’t stop once the system is implemented. Develop a plan for ongoing monitoring to assess vendor performance, security updates, and compliance throughout the contract's duration. The frequency of these reviews should align with the vendor's risk profile - higher-risk vendors demand more frequent assessments.
Keep an eye on vendor performance metrics and security updates. Notify leadership immediately if critical events occur, such as data breaches or significant leadership changes. Regularly scheduled reviews are also vital, especially when new equipment is introduced or operational processes evolve. These reviews can uncover practical risks identified by frontline staff.
Cybersecurity Risk Assessments
Cybersecurity is a top priority when working with CDSS vendors. Implement 24/7 monitoring and integrate automated workflows with your existing security systems. Conduct regular scans of vendor systems and verify certifications such as SOC 2 or HITRUST. Additionally, review their incident response plans to ensure they meet your organization's standards.
As new vulnerabilities and regulations emerge, update your security criteria to stay ahead of potential threats. This proactive approach will help protect both your organization and your patients from evolving cybersecurity risks.
sbb-itb-535baee
Using Censinet RiskOps™ for CDSS Vendor Risk Management
Managing risks associated with Clinical Decision Support Systems (CDSS) vendors requires more than occasional check-ins - it demands constant, proactive oversight. Censinet RiskOps™ steps up to this challenge by offering dynamic, real-time monitoring of rapidly changing CDSS risk factors, going beyond traditional static assessments [5]. By automating processes and incorporating continuous monitoring, the platform addresses critical risks like bias, accuracy issues, and cybersecurity threats.
One of the biggest hurdles in healthcare risk management is keeping up with the rapid pace of changes in CDSS software, especially when those changes can directly affect patient safety [5]. Manual methods simply can't keep up. Censinet RiskOps™ bridges this gap, providing tools that streamline risk evaluation while ensuring no critical detail is overlooked.
Automated Third-Party Risk Assessments
Quick and precise vendor assessments are essential, and Censinet AI™ transforms how these reviews are conducted. The platform takes lengthy security questionnaires and condenses them into clear, actionable risk summaries in just seconds. It automatically captures key documentation and integration details, eliminating the need for tedious manual processing.
This level of automation cuts review times dramatically - from weeks to mere moments. Instead of sifting through hundreds of pages of vendor documentation, risk teams can zero in on the most pressing issues, making faster and better-informed decisions about CDSS deployments.
Human-in-the-Loop Oversight
Automation is a game-changer, but human expertise remains critical, especially when patient care is on the line. Censinet RiskOps™ integrates human oversight into its workflows, ensuring that automated processes complement, rather than replace, expert decision-making.
The platform routes key findings and high-priority tasks to designated stakeholders, including members of the AI governance committee, for review and approval. This ensures that risk teams retain control over final decisions while benefiting from automation's speed and efficiency. It's a thoughtful balance - scaling processes without sacrificing safety or accountability.
Benchmarking and Continuous Monitoring
Censinet RiskOps™ also enables healthcare organizations to gauge CDSS vendor performance by comparing them across multiple healthcare delivery systems through a collaborative risk network. This benchmarking provides valuable context, helping organizations identify vendors with a proven track record of security and reliability.
The platform's command center pulls everything together in one intuitive dashboard, centralizing policies, risks, and tasks. This real-time, unified view ensures that the right teams are addressing the most pressing issues at every stage of the vendor lifecycle - from initial onboarding to ongoing monitoring after deployment. It’s a comprehensive approach that keeps oversight continuous and ensures accountability at every step.
Conclusion
Clinical Decision Support Systems (CDSS) have evolved significantly, transitioning from basic rule-based tools to sophisticated AI-driven platforms that play a critical role in patient care. However, as these systems grow more advanced, so do the risks tied to their use. Concerns such as algorithmic bias, accuracy issues, and cybersecurity vulnerabilities highlight the importance of ongoing vigilance. For healthcare organizations, ensuring the safety and reliability of CDSS must remain a top priority.
A structured, evidence-based approach to risk management is crucial for safeguarding patient safety and maintaining clinical standards. While CDSS have demonstrated their value in reducing medication errors and improving prescribing practices, their growing use has also exposed challenges like a lack of transparency and difficulties in evaluation. These issues emphasize the importance of holding vendors accountable, particularly for AI-based systems, to ensure fair and equitable outcomes for all patient populations. Rigorous evaluation of these systems, both during development and after implementation, is essential to address existing disparities and improve patient care.
Managing these risks effectively requires a proactive and continuous approach throughout the vendor lifecycle. From initial validation to ongoing monitoring after deployment, organizations must adopt strategies that go beyond manual processes or occasional reviews. Real-time monitoring, automated assessments, and human oversight must work in tandem to address unintended consequences and protect against cybersecurity threats. By implementing comprehensive risk management practices, healthcare organizations can maximize the benefits of CDSS while ensuring accuracy, fairness, and, most importantly, patient safety.
FAQs
How can healthcare organizations reduce bias in clinical decision support systems (CDSS)?
Healthcare organizations can take meaningful steps to minimize bias in Clinical Decision Support Systems (CDSS). One crucial approach is to rigorously test these systems before they are launched. This helps uncover and address potential biases early on. Beyond that, ongoing monitoring of how the system performs across different patient groups is vital to ensure both accuracy and fairness.
Incorporating diverse and representative datasets when refining algorithms is another important step. Bringing together multidisciplinary teams - including clinicians, data scientists, and ethicists - can provide varied perspectives, making the system more dependable. Additionally, being transparent about how algorithms are developed and actively working to identify and correct disparities are essential for ensuring equitable care for all patients.
How can we ensure the accuracy of Clinical Decision Support Systems?
To ensure Clinical Decision Support Systems (CDSS) remain accurate, it's crucial to emphasize data validation at the point of entry. Incorporating automated error-checking mechanisms can catch mistakes early, while AI tools can help process unstructured data more efficiently. Regular data quality audits and smooth integration with electronic health records (EHRs) are also key to maintaining system reliability.
Equally important is providing thorough staff training to help users understand and effectively operate the system. By continuously monitoring and improving workflows, organizations can uphold accuracy and reliability standards, which directly contribute to enhancing patient safety.
How does Censinet RiskOps™ improve cybersecurity for managing CDSS vendors?
Censinet RiskOps™ enhances cybersecurity for clinical decision support systems (CDSS) by offering continuous risk assessments, real-time risk monitoring, and automated response plans. These tools work together to quickly pinpoint and resolve potential vulnerabilities, helping to prevent security breaches before they occur.
By simplifying data sharing and cutting down on manual workflows, Censinet RiskOps™ enables organizations to prioritize proactive risk management. This not only boosts operational efficiency but also supports safer outcomes for patients.
Related Blog Posts
- Clinical Decision Support System Vendor Risk: Bias, Accuracy, and Patient Safety
- How Vendor Failures Impact Patient Outcomes: Real-World Healthcare Case Studies
- Healthcare Predictive Analytics Vendor Risk: Data Quality and Model Reliability
- The Healthcare AI Paradox: Better Outcomes, New Risks
{"@context":"https://schema.org","@type":"FAQPage","mainEntity":[{"@type":"Question","name":"How can healthcare organizations reduce bias in clinical decision support systems (CDSS)?","acceptedAnswer":{"@type":"Answer","text":"<p>Healthcare organizations can take meaningful steps to minimize bias in Clinical Decision Support Systems (CDSS). One crucial approach is to rigorously test these systems before they are launched. This helps uncover and address potential biases early on. Beyond that, ongoing monitoring of how the system performs across different patient groups is vital to ensure both accuracy and fairness.</p> <p>Incorporating <strong>diverse and representative datasets</strong> when refining algorithms is another important step. Bringing together multidisciplinary teams - including clinicians, data scientists, and ethicists - can provide varied perspectives, making the system more dependable. Additionally, being transparent about how algorithms are developed and actively working to identify and correct disparities are essential for ensuring equitable care for all patients.</p>"}},{"@type":"Question","name":"How can we ensure the accuracy of Clinical Decision Support Systems?","acceptedAnswer":{"@type":"Answer","text":"<p>To ensure Clinical Decision Support Systems (CDSS) remain accurate, it's crucial to emphasize <strong>data validation</strong> at the point of entry. Incorporating automated error-checking mechanisms can catch mistakes early, while AI tools can help process unstructured data more efficiently. Regular data quality audits and smooth integration with electronic health records (EHRs) are also key to maintaining system reliability.</p> <p>Equally important is providing <strong>thorough staff training</strong> to help users understand and effectively operate the system. By continuously monitoring and improving workflows, organizations can uphold accuracy and reliability standards, which directly contribute to enhancing patient safety.</p>"}},{"@type":"Question","name":"How does Censinet RiskOps™ improve cybersecurity for managing CDSS vendors?","acceptedAnswer":{"@type":"Answer","text":"<p>Censinet RiskOps™ enhances cybersecurity for clinical decision support systems (CDSS) by offering <strong>continuous risk assessments</strong>, <strong>real-time risk monitoring</strong>, and <strong>automated response plans</strong>. These tools work together to quickly pinpoint and resolve potential vulnerabilities, helping to prevent security breaches before they occur.</p> <p>By simplifying data sharing and cutting down on manual workflows, Censinet RiskOps™ enables organizations to prioritize proactive risk management. This not only boosts operational efficiency but also supports safer outcomes for patients.</p>"}}]}

